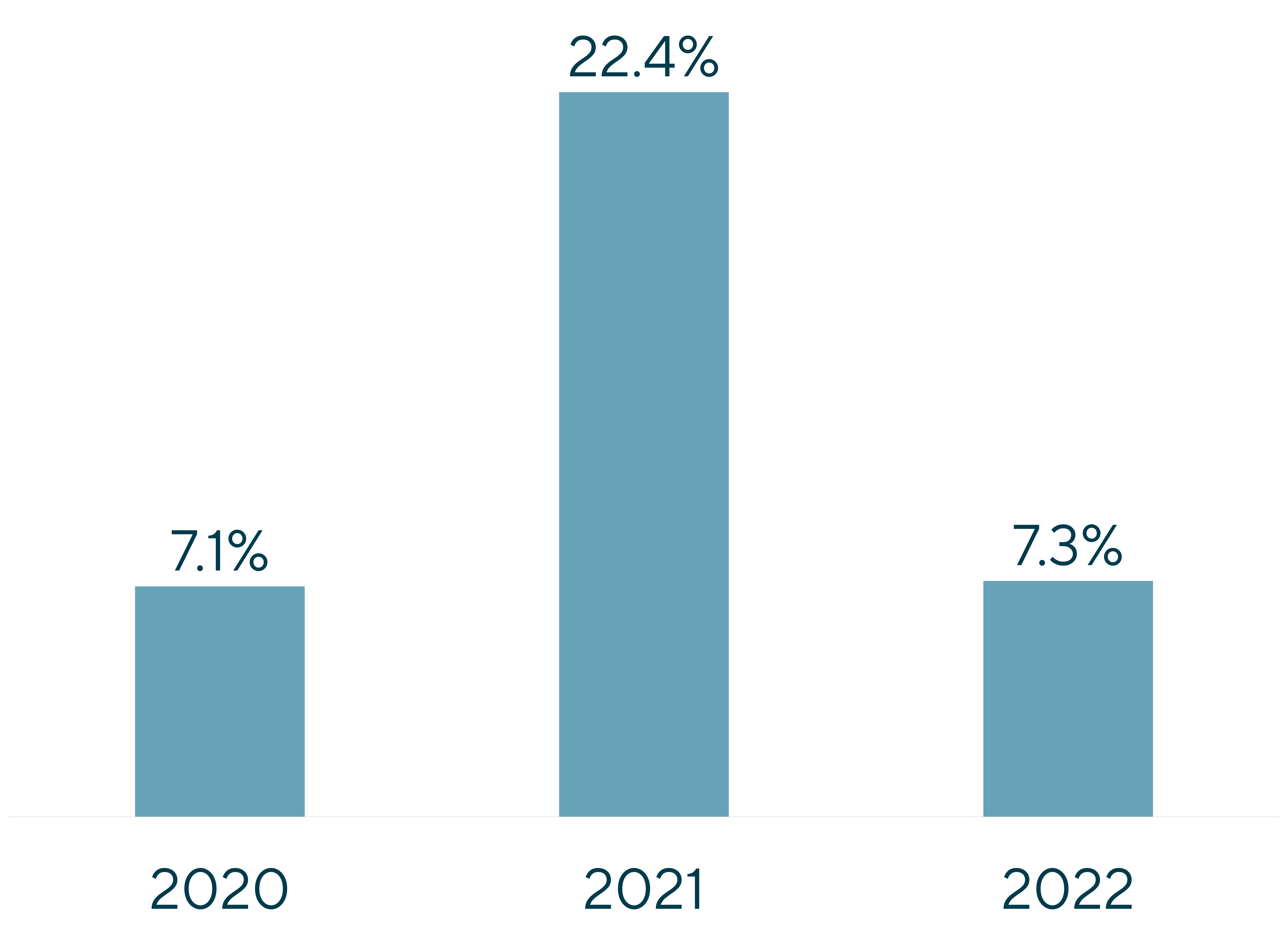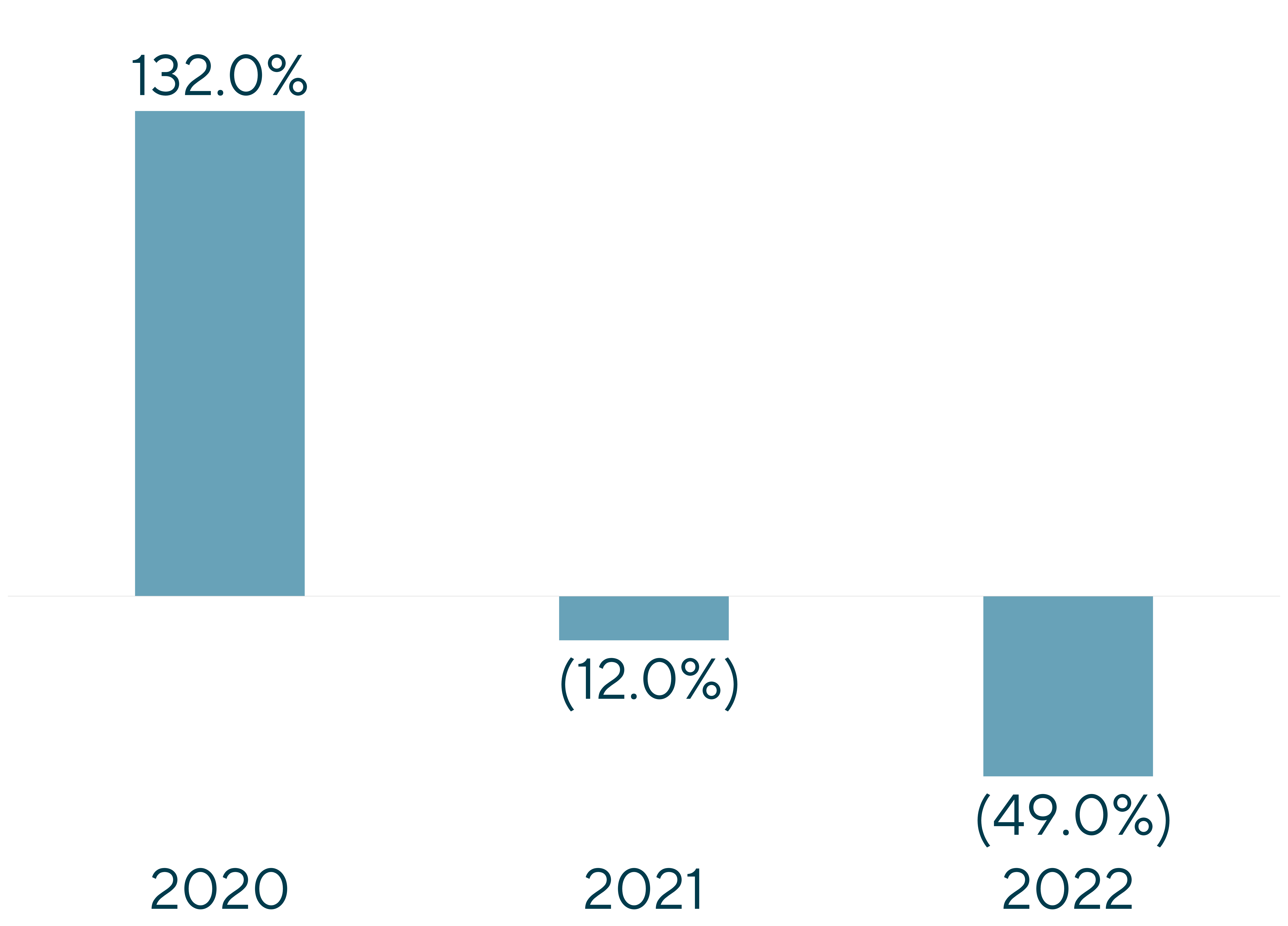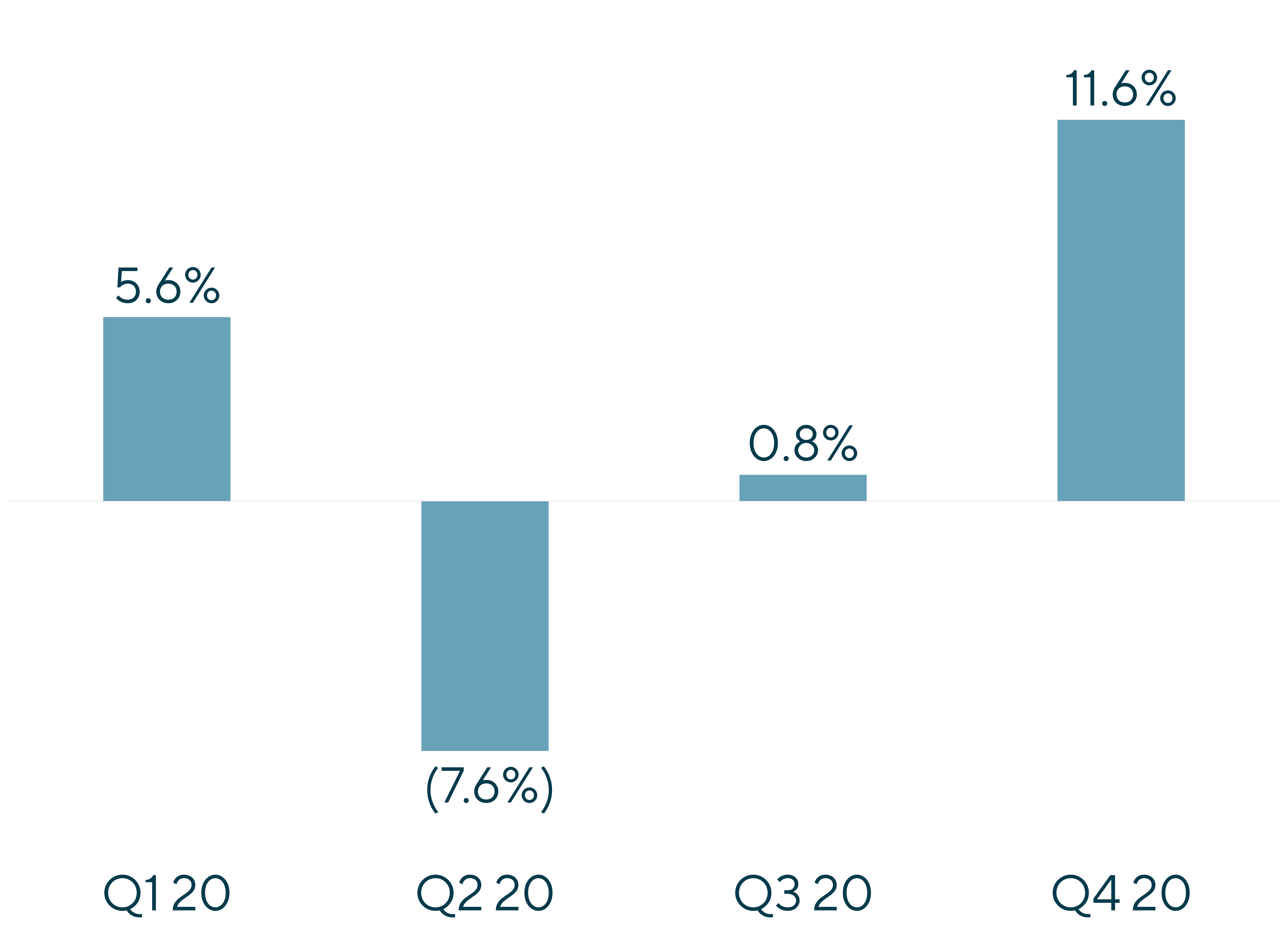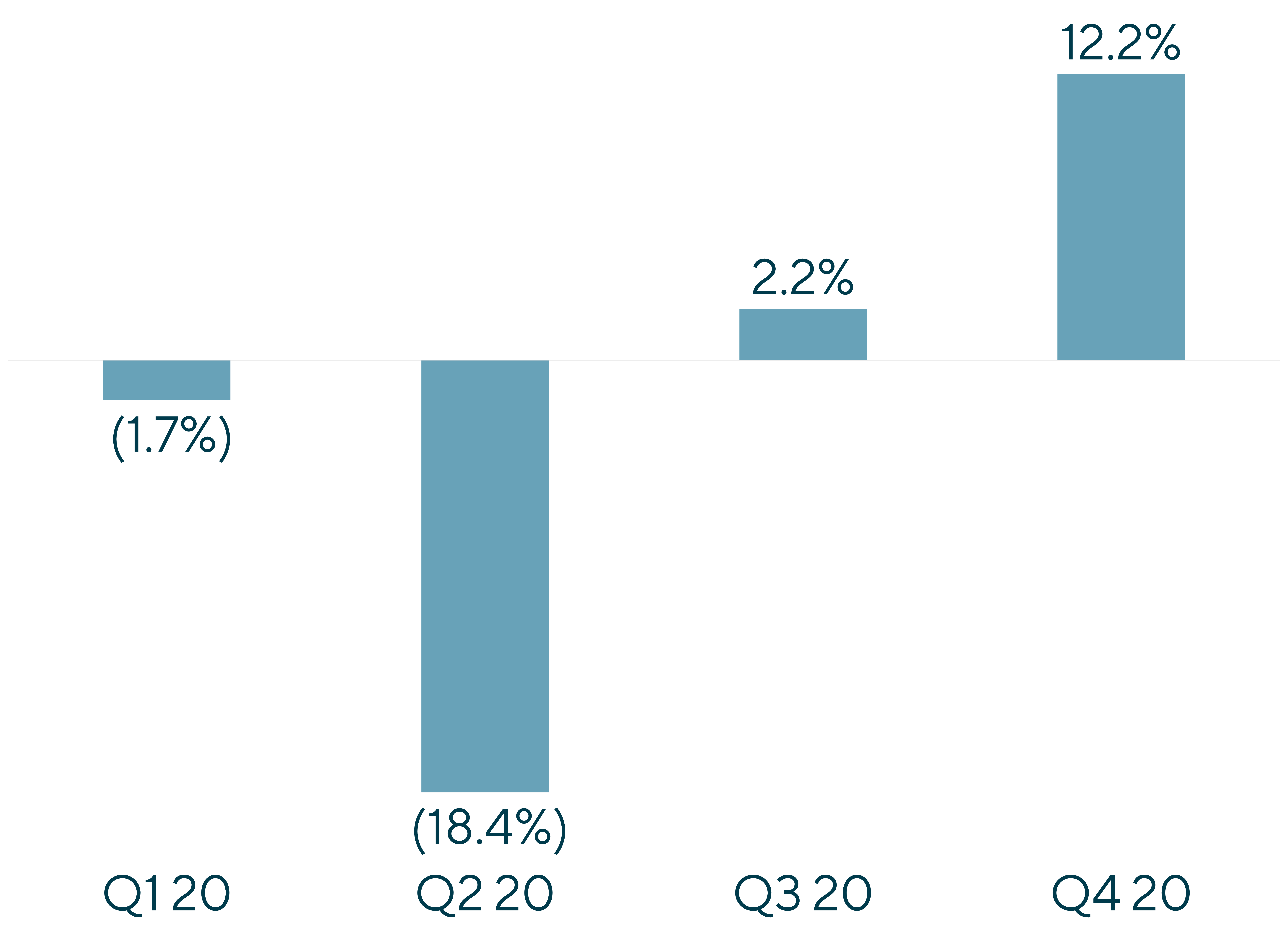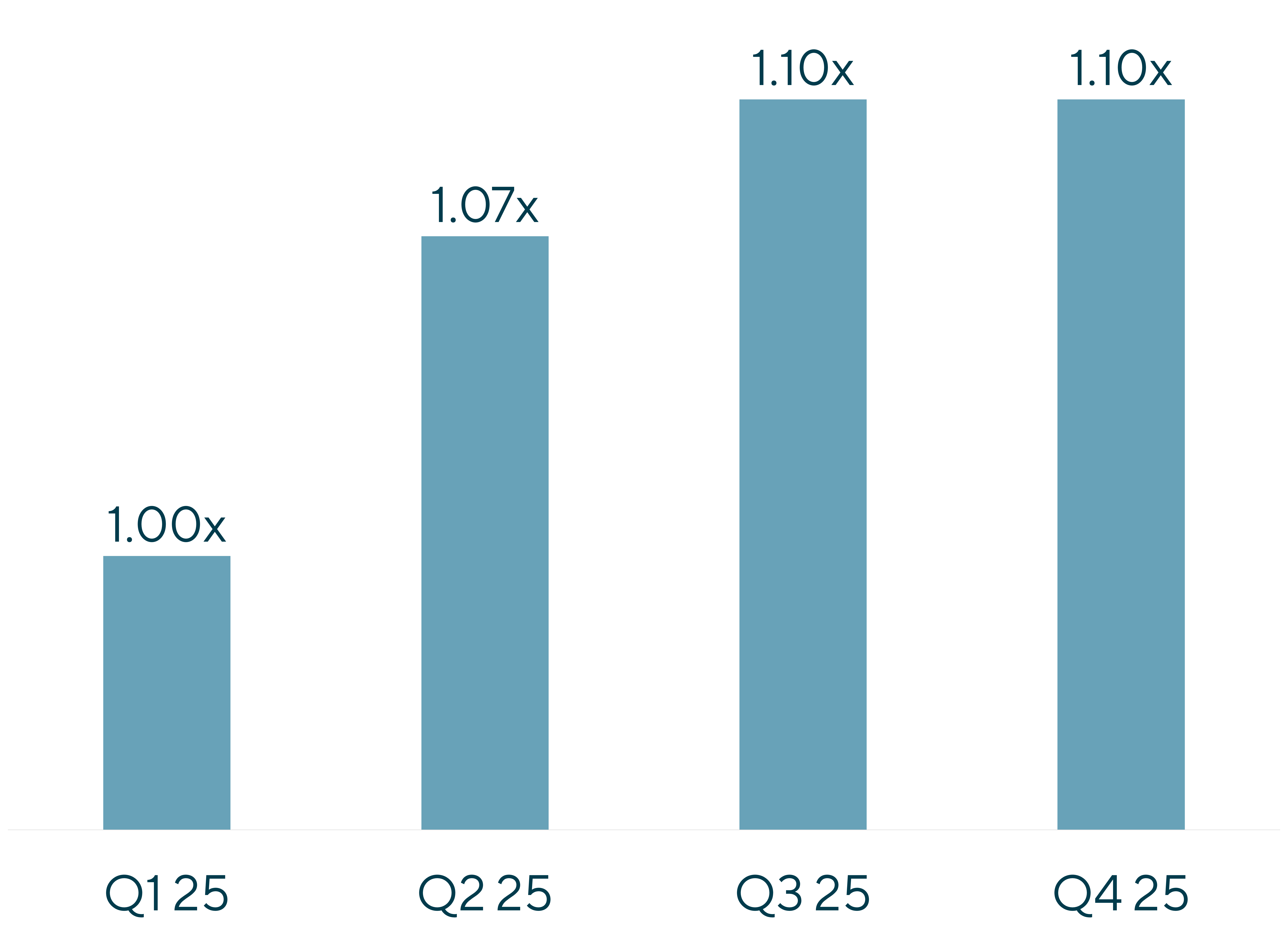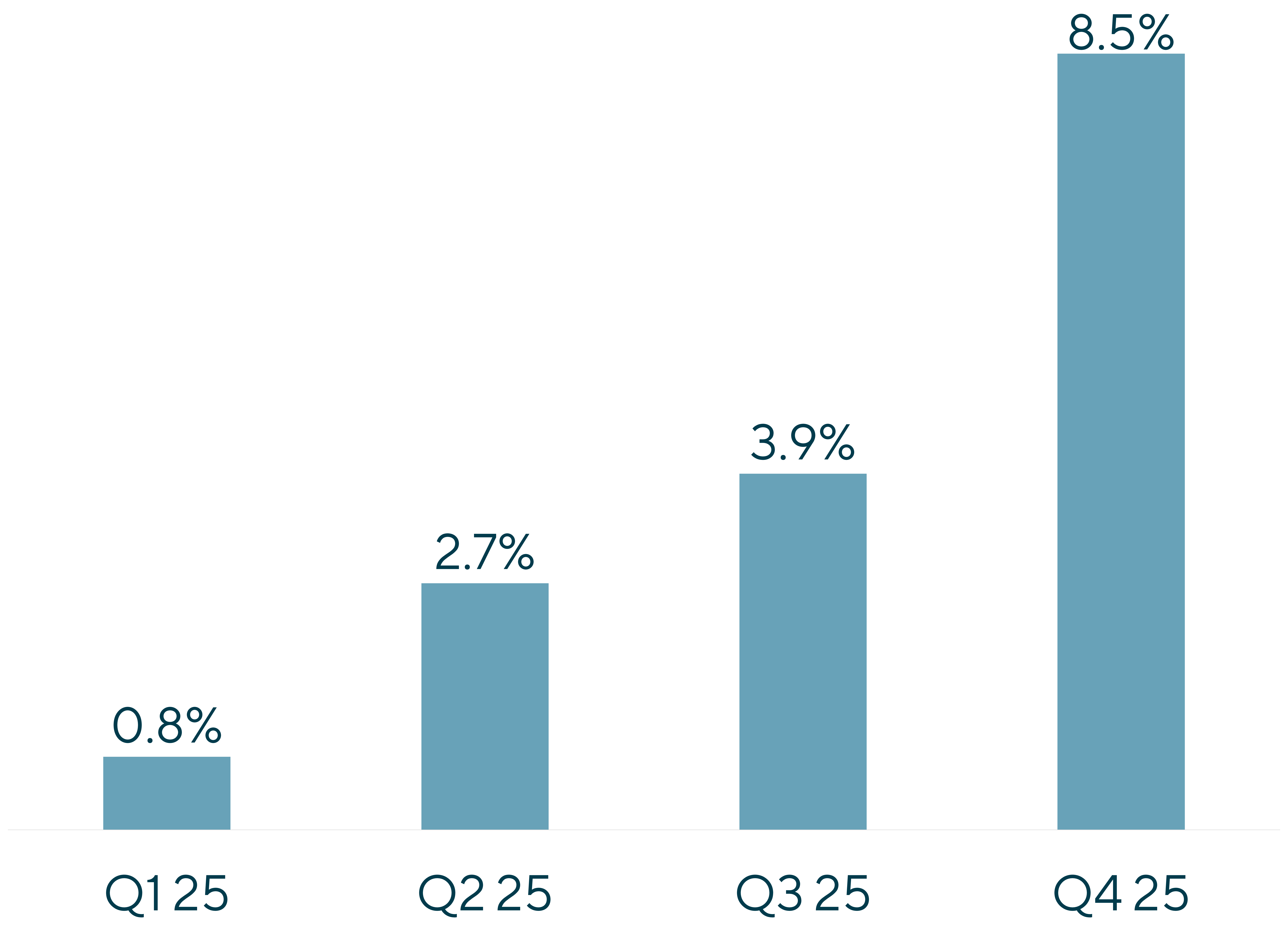Pharma Services Deep Dive: From Post-Pandemic Reset to the Next Cycle
Introduction: Experiencing a Primarily Cyclical Shift, not a Structural One
Over the past several years, the clinical trial services market has experienced one of the most pronounced demand cycles in its history. What felt like structural acceleration at the time, particularly in the windfall of 2021, now appears to be far more nuanced: a funding-driven, lagged demand cycle, with meaningful implications for how investors are approaching the sector now and in the future.
This clear distinction should matter to potential investors, as structural growth narratives potentially justify sustained, elevated multiples for assets within the sector. Cyclical dynamics, however, particularly those tied to external funding conditions, demand a more disciplined, timing-sensitive investment thesis.
From the COVID-induced disruption in 2020, through the demand surge in 2021, and into the normalization and eventual contraction observed in 2023 and 2024, the clinical trial services landscape appears to be coming full circle, albeit somewhat slowly. As 2026 nears the midway point, early signs of stabilization are beginning to emerge, even if public and private markets do not fully reflect this reality, but the nature of this recovery, and who ultimately benefits, remains one of the questions Lincoln International’s Healthcare Group receives most frequently.
Summary
-
The clinical trial services landscape is showing signs of stabilization and recovery, and Lincoln International’s Healthcare Group analyzes who will benefit and what is driving the growth.
- Sign up to receive Lincoln's perspectives
The Core Framework: Misunderstood, Misinterpreted or Overlooked Demand Models?
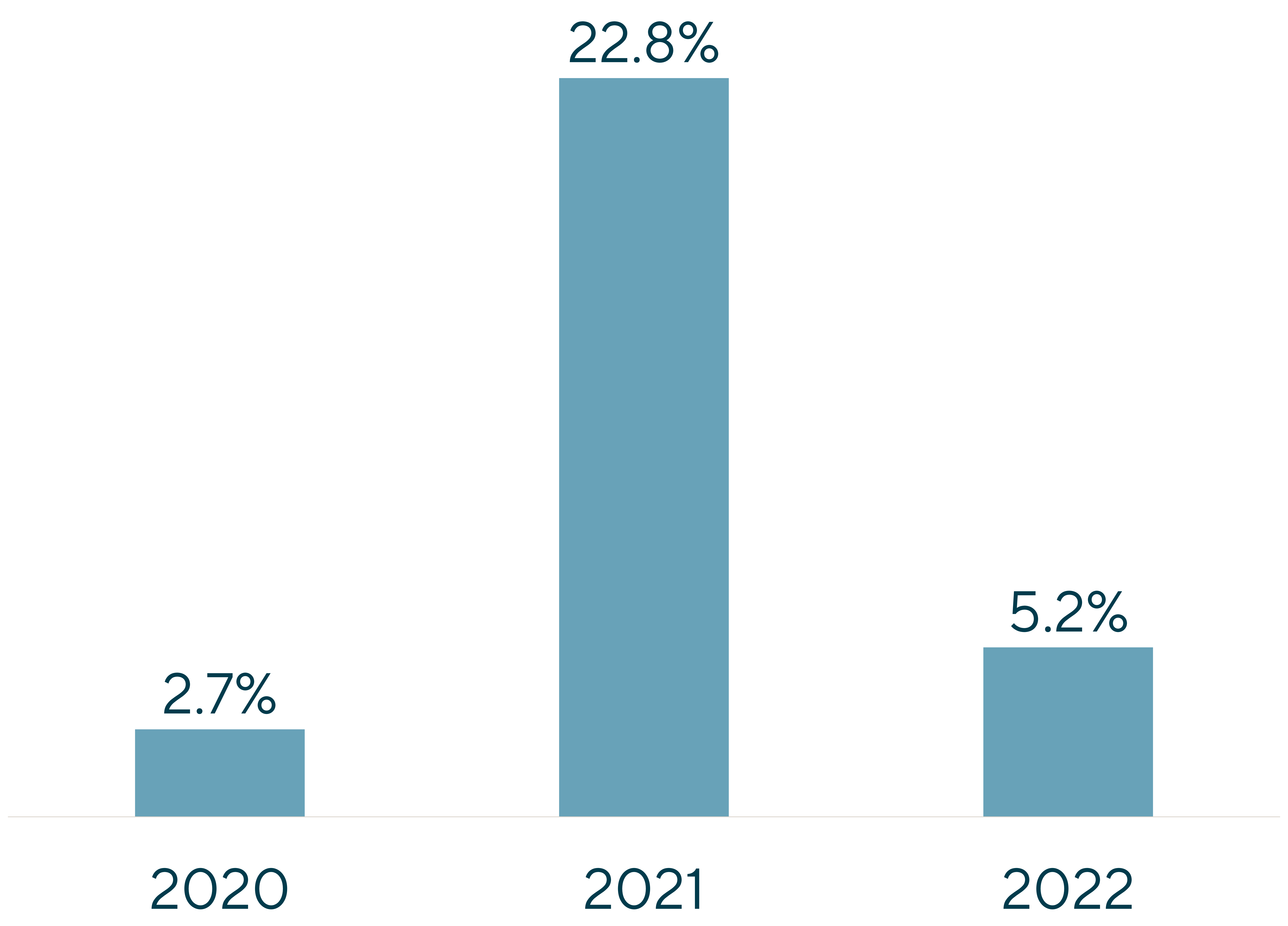
At its core, pharma services companies servicing clinical trial activities are governed by a simple model of funding, which drives bookings, which translates to backlog, which is eventually recognized as revenue. This lagged demand model within the sector is best exhibited by growth that remained strong well into 2022 despite the continued weakening of the funding environment and the deceleration of bookings growth year-over-year.
| Exhibit 1: Annual Revenue Growth (YoY) | Exhibit 2: Annual Bookings Growth (YoY) | Exhibit 3: Annual Biopharma Funding Growth (YoY) |
Using bookings as a strong proxy for the future operating environment shows that slowing demand was not fully visible until late 2023, and demand contraction did not become visible until 2024. Seasoned investors understood the cyclical nature of this sector, and while continued organic revenue growth was always a strong component of investment theses, scrutiny over bookings and backlog (as well as conversion rates) painted a clearer picture of lagged strength vs. real-time demand and the ultimate sustainability of future growth.
An Unprecedented Cycle: How Did We Get Here?
2020: Disruption but a Demand Opportunity
As with most industries, the onset of the pandemic brought significant disruption to the entirety of clinical development activities. Trials, both ongoing and new, slowed to a crawl as patients were either unable to, or chose not to, participate in studies, and there were severe operational constraints on the physical site locations. Budgets, and what funds should or should not be allocated towards, became the focus of every trial sponsor. As a result, revenue and EBITDA declined sharply by -7.6% and -18.4%, respectively, YoY in Q2 2020 before a quick correction occurring in Q4 2020.
| Exhibit 4: Quarterly Revenue Growth (YoY) | Exhibit 5: Quarterly EBITDA Growth (YoY) |
What at the time appeared as an unprecedented demand shock was ultimately joined by a severe timing dislocation in the market, with several factors building to support this trend, including:
| Trials being delayed though not yet cancelled |
| Continued outsourcing rates as sponsors sought flexibility in uncertain times |
| Rapid and unprecedented mobilization of COVID-related development programs |
The combination of these factors drove strong backlog growth into the end of the year even as revenue lagged, resulting in strong book-to-bill ratios that provided early evidence that future demand was not impaired but rather being reprioritized or deferred.
2021: Demand Shock vs. Structural Growth
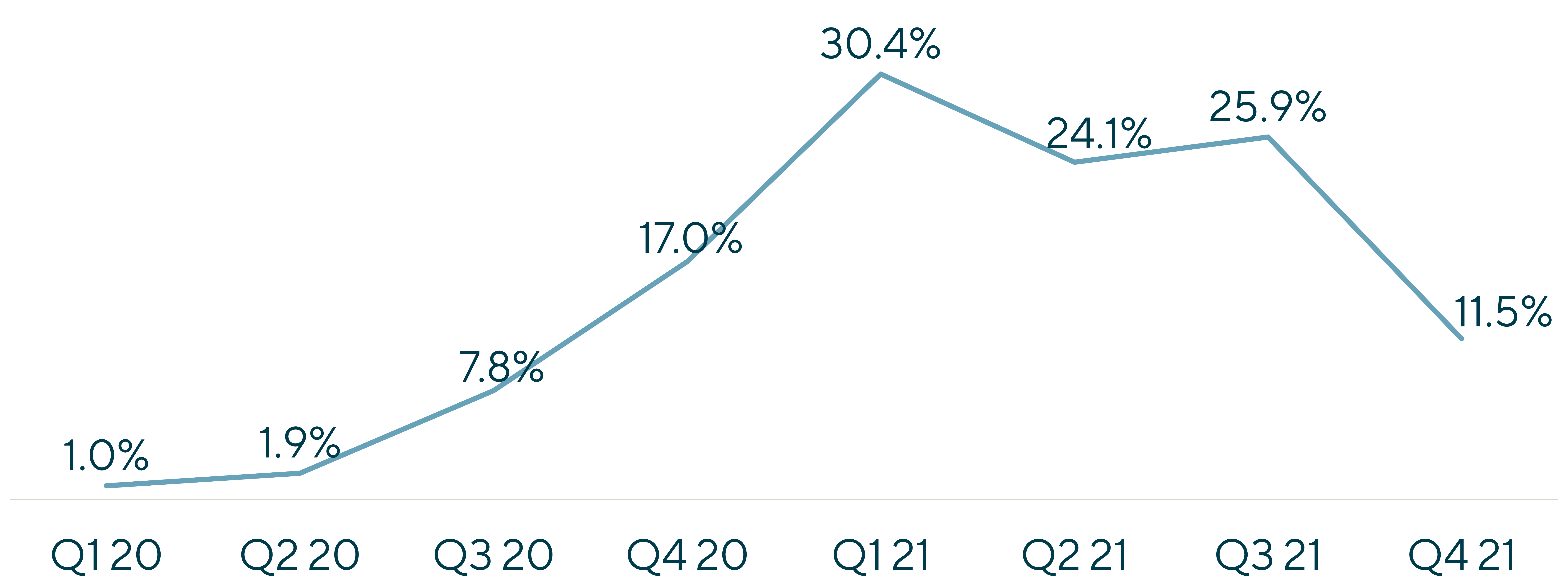
2021 saw demand acceleration at an arguably unsustainable degree, aided by trial activity rebounding, continued tailwinds tied to a robust biotech funding environment and the continued proliferation of COVID-related programs which converted from backlog to revenue rapidly.
As a result, bookings growth exploded at 22% throughout the year (though off soft comparable data), and backlogs continued to expand. Importantly, this backlog expansion was paired with elevated conversion rates as quarterly backlog conversion as a percentage of prior period backlog rebounded in 2021 compared to 2020, largely driven by the warp-speed efforts in COVID vaccine related programs.
| Exhibit 6: Quarterly Backlog Conversion | Exhibit 7: Quarterly Bookings Growth (YoY)
|
When evaluating opportunities in the sector, investors continued to pressure test whether these trends, even with some level of haircutting, represented a new steady state and what level, if any, of this was pull-forward of future demand. Sponsors, flush with capital and operating with urgency, accelerated trial initiation timelines, and programs that may have staggered previously were launched in rapid succession. With the ongoing funding spigot, sponsor pipelines expanded rapidly and looked to have no end in sight, delaying the need to prioritize, optimize or rationalize. All of this set the stage for a future “air pocket” driven by shifting dynamics in the funding markets and construction of drug development pipelines.
2022: A Transitioning Market Signals False Stability
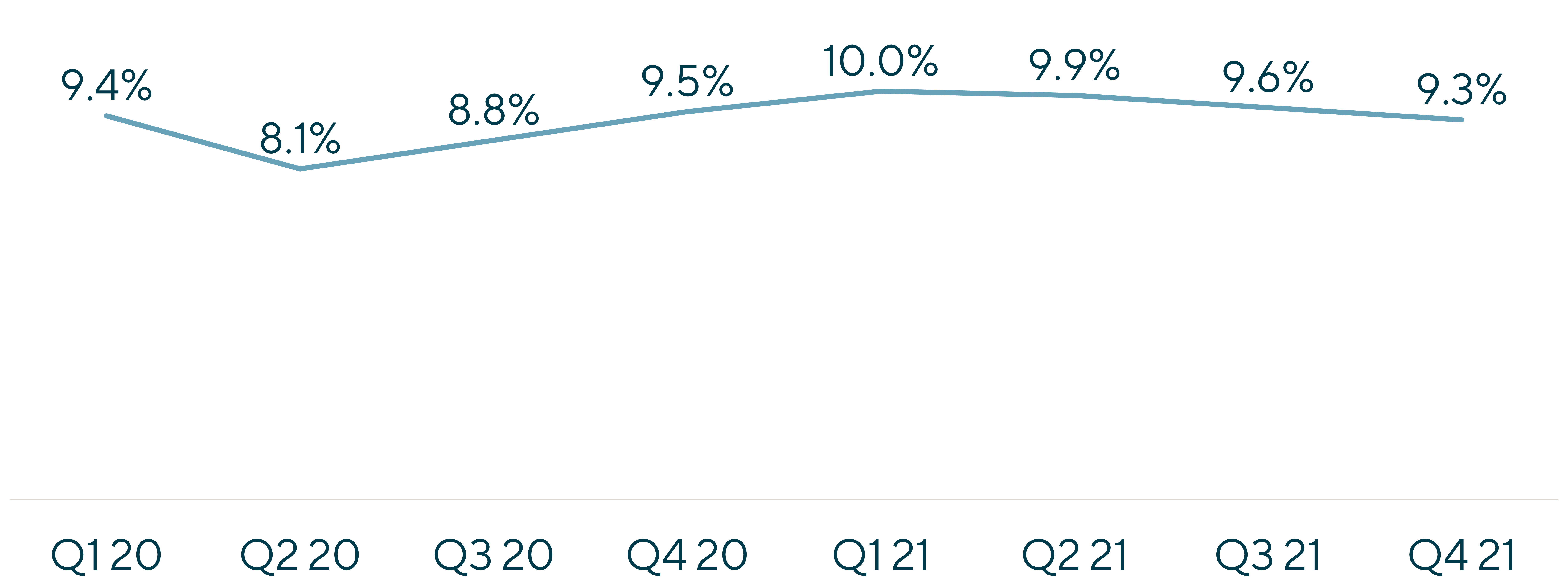
The sector was a highly favored area for capital deployment coming out of 2021. Bookings growth remained robust through mid-2022, and EBITDA margin expansion continued as pricing power held for the sector in the face of this unprecedented demand.
However, initial signs began to emerge of a softening market. The bookings growth that had driven such a robust environment began to decelerate meaningfully, and levels of backlog conversion began to contract. This coincided with the beginning of a tighter funding environment and a shift towards more disciplined capital allocation by trial sponsors, including portfolio optimizations and reorganizations. These factors, combined with excess demand working its way through the sector and a slowing rate of new programs being greenlit, led the way to a multi-year digestion phase within the sector.
| Exhibit 8: Quarterly Backlog Conversion Rate Growth (YoY) |
2023 – 2024: The Great Reset
The biotech bull market that peaked in 2021 and declined sharply had come to a head, as the nature of the lagged demand model of the sector became visible in 2023 and intensified into 2024. Capital market dependent (“CMD”) biotech had been a robust source of demand for pharma services companies broadly and a primary driver of growth in the sector, but the true nature of a finite capital pool resulted in several painful headwinds:
| Decreasing number of clinical trial starts |
| Portfolio optimization and reorganization (i.e., pharma was culling its shotgun approach and prioritizing its best programs) |
| Widespread cost containment and discussions of insourcing vs. outsourcing |
| Sponsors exiting therapeutic areas entirely at the risk of being underweight in key emerging ones |
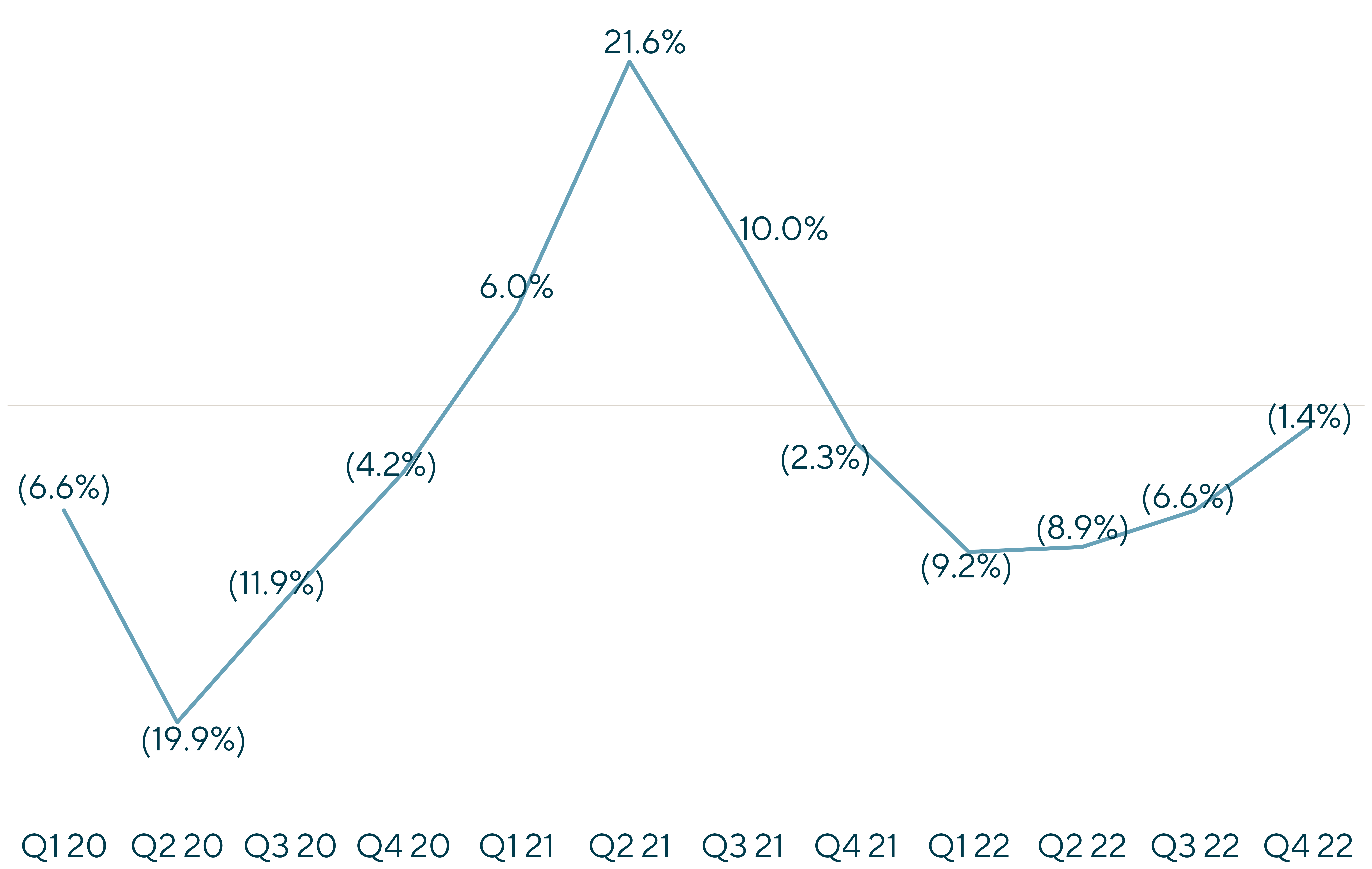
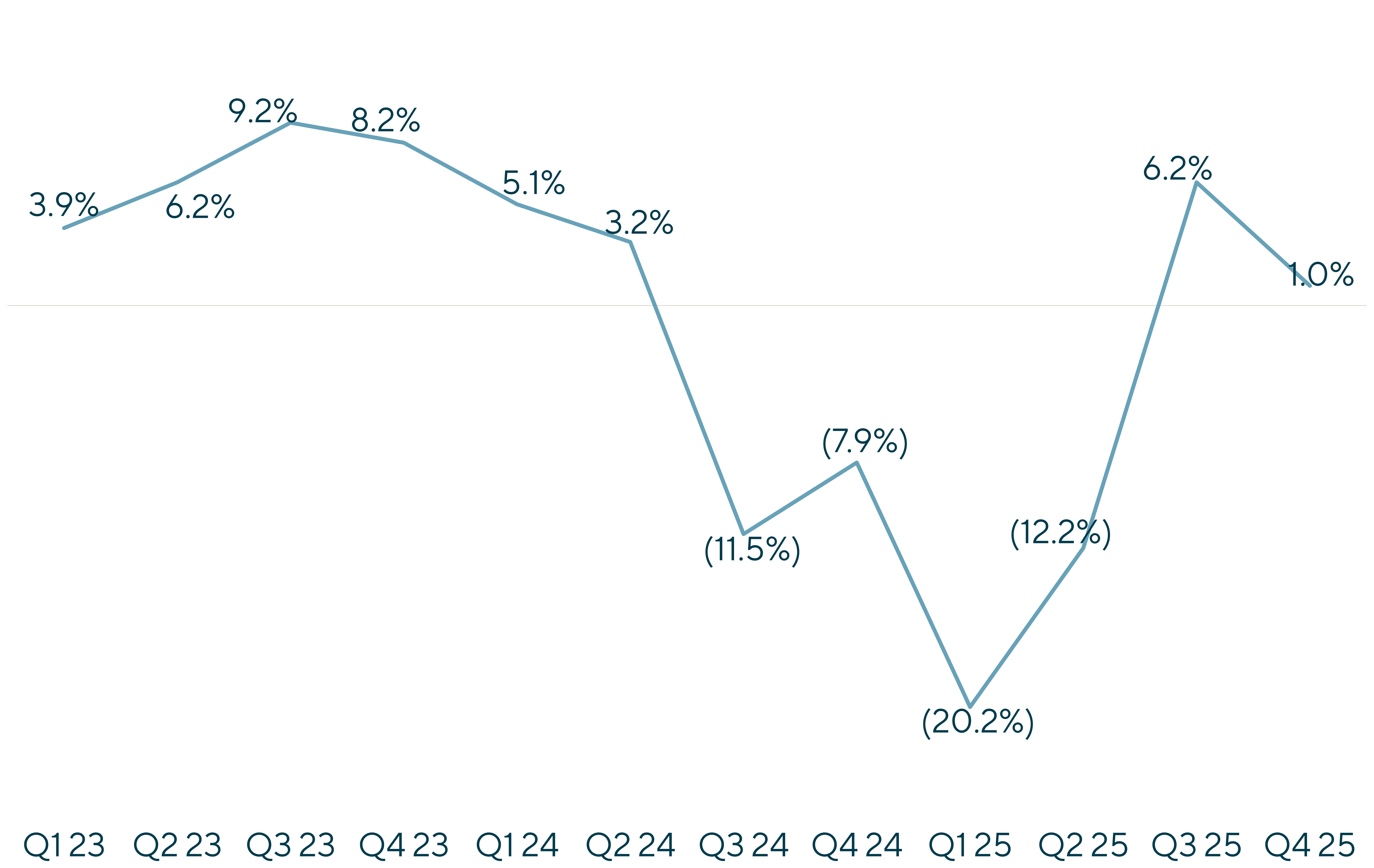
While revenue continued to remain resilient initially, supported by record backlogs, it was now decelerating sharply and reaching trough levels. The confluence of events that had occurred in the sector since the start of the pandemic resulted in what can best be described as a true demand reset as 2024 came to close. New bookings growth turned decisively negative, backlog growth steadily deteriorated and book-to-bill compressed close to a zero-growth rate scenario.
| Exhibit 9: Quarterly Bookings Growth (YoY) |
2025: Stabilization and the Start of a Recovery
2025 proved once again the cyclical nature of the industry and that, regardless of how dynamic a sector is, positive reversion can sometimes be slow in the beginning with false starts. However, several indicators that will underpin a recovery showed significant improvements, including:
| Bookings saw positive year-over-year growth in the second half of the year |
| Book-to-bill ratios recovered from their lows of 1.0x, pointing to signs of a better future operating environment |
| Revenue remained resilient and ticked upward compared to 2024 |
| The biotech funding environment appeared more constructive, if not more selective |
| Portfolio optimization is no longer capturing extensive headlines |
Given the lessons learned in the prior years, investors were still wise to view these positives as representative of stabilization from a low base, the inverse of what was seen in 2021 compared to 2020. The contrarian viewpoint would be to point to backlog growth remaining muted, new trial starts flat to pre pandemic levels, biotech funding remaining choppy, and the emerging threat of Chinese contract research organizations. However, the cyclical nature of the sector is most likely to benefit those who take a patient, longer-term view or approach, which has the potential to pay significant dividends to more patient investors.
| Exhibit 10: Quarterly Book-to-Bill | Exhibit 11: Quarterly Revenue Growth (YoY) |
2026 and Beyond: What Does This All Mean?
The past several years highlighted the volatility that can occur in a cyclical sector such as pharma services companies addressing clinical trial needs; however, it has also brought to light several areas of structural strength and differentiation which make the sector such a compelling investment.
|
1 |
Flexibility is highly valued: Nimble platforms that can seamlessly pivot toward emerging models and solutions are well-positioned to capture the increased need for flexibility from partners that sponsors select in valuable clinical trials |
|
2 |
Specialization is a rewarded differentiator: As sponsors look to drive greater efficiencies in bringing their products to market, partners who can provide unique capabilities or insights throughout R&D in high-demand therapeutic areas and service lines will be market share takers |
|
3 |
Complexity is only increasing: Novel modalities, protocol design, and data requirements alone disproportionately benefit those partnering with sponsors on their clinical trial execution who understand the dynamic changes occurring throughout the clinical trial landscape in real-time |
|
4 |
Operational resilience remains: Despite all the noise over the past few years, from slowing growth to funding dynamics, many businesses addressing clinical trial activities have demonstrated sustained or expanding EBITDA margins highlighting operational discipline |
|
5 |
Longer horizon of R&D funding shows the full picture: Despite the COVID- and capital markets-driven spike and subsequent contraction in biotech funding, total biotech funding has grown at a 6% CAGR since 2019 |
|
6 |
Subsector-driven growth in pharmaceutical R&D: A continued focus on bringing blockbuster drugs to market in key therapeutic areas, such as oncology, obesity & metabolic disorders and neurodegenerative, presents significant opportunities to drive growth across the clinical trial spectrum |
|
7 |
Numbers driving change: There is an increasing demand for differentiated data and analytical offerings which can shape the future of more efficient trial execution and proprietary offerings provide a significant competitive moat, particularly in the face of potential AI disruption |
These factors, taken in totality, underscore the intrinsic attractiveness of platform investments in assets serving the clinical development pathway addressing the needs of sponsors across a range of clinical trial activities.
How Should Investors Prioritize Facets of the Next Stage of the Cycle?
While the elements in this issue provide a highly informative backdrop to the pharma services sector from an investing perspective, expanding the aperture beyond services focused on clinical trials highlights a broader shift across the pharma services value chain, many of which do mirror the dynamics across clinical development. While there are signs of sector recovery being already underway, it is likely to benefit, perhaps disproportionately, assets that combine deep specialization with flexibility in the delivery of their solutions. In contrast, highly discretionary and undifferentiated services (i.e. non-mission-critical) remain more exposed to continued budgetary scrutiny, uneven demand recovery and the overall cyclicality being observed.
Lincoln believes that the next phase of the sector will be defined not by a rising tide like in 2021, but by greater levels of dispersion with the biggest drivers being the aforementioned focus on flexibility and specialization. Investors focusing on assets combining these features, or highly proficient in one or the other, will be best positioned to capture a disproportionate share of the recovery. More commoditized and “one size fits all” solutions will lag regardless of how quickly or strong the demand environment becomes. A useful framework to help formulate potential investment theses is highlighted by Lincoln’s categorical benchmarking below:
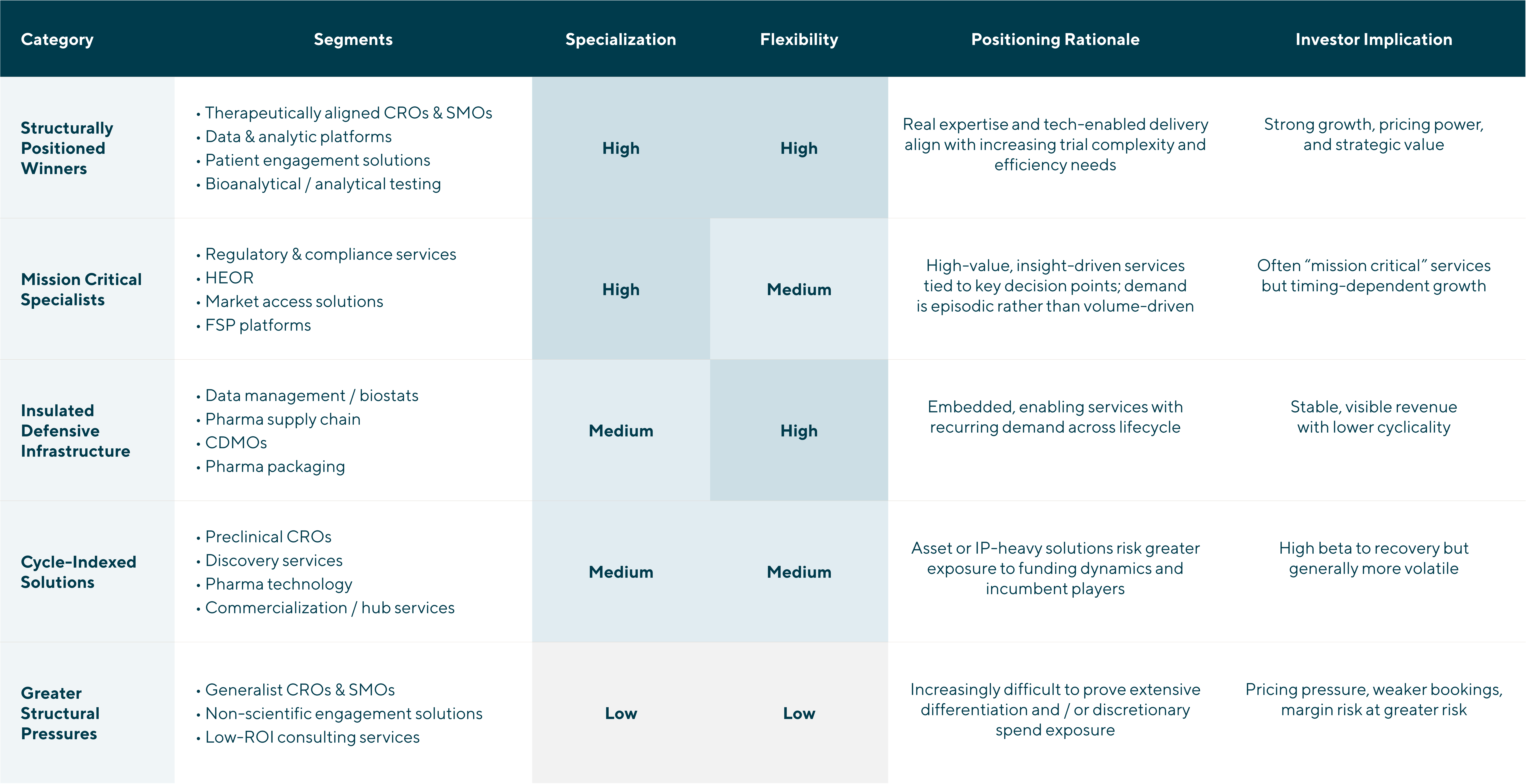
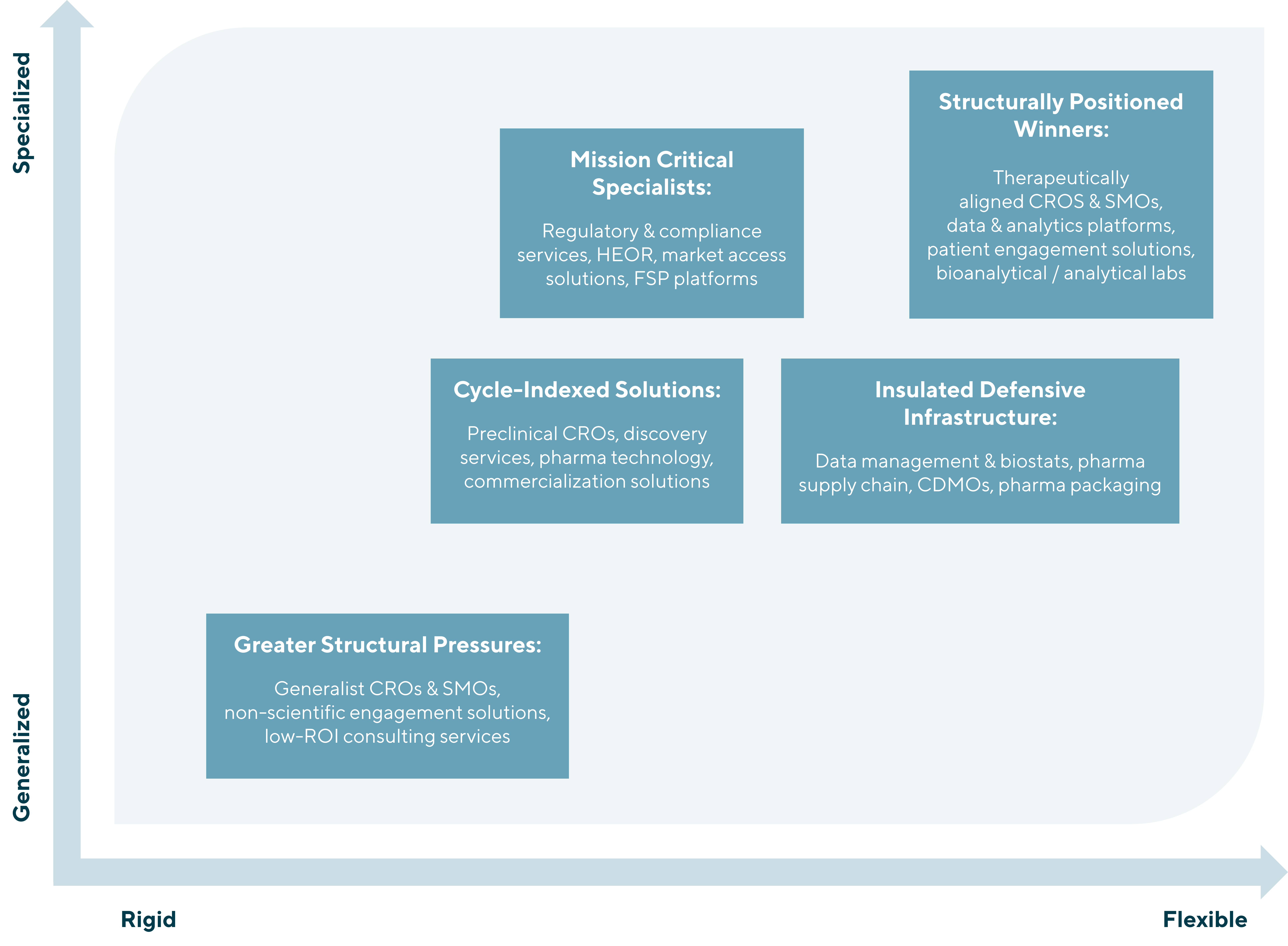
This analysis serves as the foundation for an ongoing series, in which Lincoln’s experts will further explore how investors can evaluate opportunities across the pharma services landscape. Future installments will dive deeper into the team’s framework for assessing subsector dynamics, identifying relative winners and losers and underwriting investments through the next stage of the cycle.
We look forward to sharing additional perspectives and welcome the opportunity to connect on how these trends and considerations impact current opportunities in the sector. Reach out to the professionals below to discuss how you can leverage the sector’s momentum and growth path.
Contributors

I am inspired by partnering with companies that are enabling innovation across the pharmaceutical continuum. Advising organizations helping to accelerate the discovery, development and delivery of therapies that change patients' lives provides a deep sense of purpose.
Don Matz
Managing Director
New YorkMeet Professionals with Complementary Expertise in Healthcare

It’s extremely rewarding to work in one of the largest and most diverse global business sectors helping support clients to realize their goals.
Matthew Lee
Managing Director, Head of UK & Co-Head of Healthcare, Europe
London
I enjoy working closely with clients to overcome challenging situations and to develop strategies to meet their business goals.
Dirk-Oliver Löffler
Managing Director & Co-Head of Healthcare, Europe
Frankfurt
I am inspired by partnering with companies that are enabling innovation across the pharmaceutical continuum. Advising organizations helping to accelerate the discovery, development and delivery of therapies that change patients' lives provides a deep sense of purpose.
Don Matz
Managing Director
New York
My goal is to bring the best of Lincoln to each and every transaction, ensuring the topmost outcomes for our clients.
Roderick O’Neill
Managing Director & Global Head of Healthcare
New York